which was first identified by an outbreak of respiratory illness cases in Wuhan City, Hubei Province, China. It was initially reported to the WHO on December 31, 2019.The global COVID-19 crisis has had and will continue to have a catastrophic impact on each person globally. By over 266 million diagnosed cases globally, the COVID-19 pandemic is a worldwide health emergent issue affecting all human’s life globally. Since pandemic started various medicines have been evaluated and many of them have been prescribed for the treatment of COVID-19 despite the conflicting or preliminary results of the clinical trials. Both Favipiravir and Molnupiravir impairs SARS-CoV-2 replication, Favipiravir does it by selectively inhibiting RNA polymerase and Molnupiravir does it by increasing the frequency of viral RNA mutations. One of the distinguishing points of both of them is that they are an oral antiviral drug designed to treat COVID-19, which make them more convenient treatment options for patients. Also, on 30th December, U.S FDA panel of advisory narrowly endorses Molnupiravir for COVID-19 treatment.
Hence, one of Parsian’s fields of activity is research and development in COVID-19 to manufacture active pharmaceutical ingredients (API) and Granules of Favipiravir and Molnupiravir with the highest global standards to help patients to be cured in a better way by the latest guidelines and with the lowest adverse effects.
Covid 19
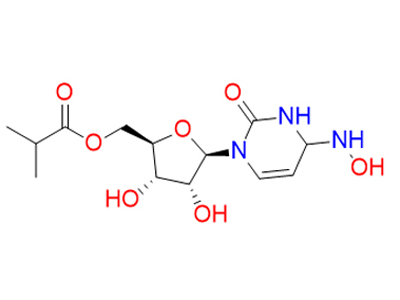
Cas No: 2349386-89-4
Mechanism Of Action: Molnupiravir is hydrolyzed in vivo to N4-hydroxycytidine, which is phosphorylated in tissue to the active 5’-triphosphate form, and incorporated into the genome of new
Covid 19
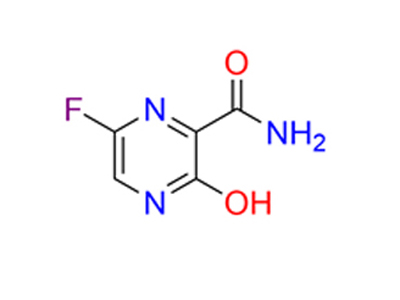
Cas No: 259793-96-9
Mechanism Of Action: The active favipiravir-RTP selectively can inhibit RNA polymerase and prevent replication of the viral genome. There are several hypotheses as to how