Mechanism Of Action:
The B cell receptor (BCR) pathway regulates lots of cellular processes including, proliferation, differentiation, and apoptosis which are essential for the functioning and survival of both normal and malignant B cells. In B cell malignancies, such as CLL; aberrant BCR signaling plays a critical role in the pathogenesis of disease. The BCR pathway is responsible for the phosphorylation of numerous protein tyrosine kinases (PTKs), including Lyn, SYK, and Bruton’s tyrosine kinase (BTK). These PTKs showed constitutively and over-expressed activity in CLL, which leads to uncontrolled proliferation and survival of malignant B cells, so, there has been rapid clinical development of inhibitors targeting these PTKs. Among the many PTKs involved in BCR signaling, BTK, a tyrosine kinase member of the Tec kinase family, is a distinctive therapeutic target. Upon BCR activation, BTK becomes activated by other PTKs, such as Lyn and SYK, resulting in activation of downstream transcription factors necessary for B cell proliferation and differentiation.
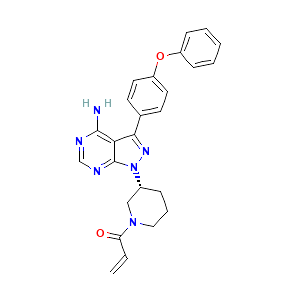
Ibrutinib is a highly potent, selective, and irreversible small-molecule inhibitor of BTK. It forms a covalent bond with a cysteine residue (CYS-481) at the active site of BTK, leading to inhibition of BTK enzymatic activity. It can also abrogate the full activation of BTK by inhibiting its auto phosphorylation at Tyr-223. This inhibition prevents downstream activation of the BCR pathway and subsequently blocks cell growth, proliferation, and survival of malignant B cells.
Indication:
- Mantle cell lymphoma (MCL) who have received at least one prior treatment
- Chronic lymphocytic leukemia (CLL)/Small lymphocytic lymphoma (SLL)
- Chronic lymphocytic leukemia (CLL)/Small lymphocytic lymphoma (SLL) with 17p deletion
- Waldenström’s macroglobulinemia (WM)
- Marginal zone lymphoma (MZL) who require a medicine by mouth or injection (systemic therapy) and have received a certain type of prior treatment
- Chronic graft versus host disease (cGVHD) after failure of 1 or more lines of systemic therapy.