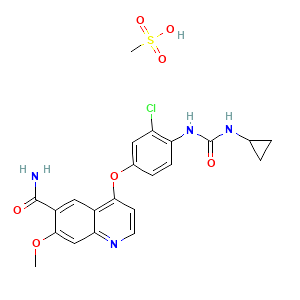
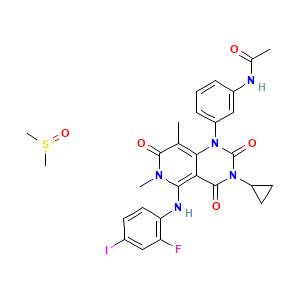
Mechanism Of Action:
The active favipiravir-RTP selectively can inhibit RNA polymerase and prevent replication of the viral genome. There are several hypotheses as to how favipiravir-RTP interacts with RNA dependent RNA polymerase (RdRp). Some studies have demonstrated that when favipiravir-RTP is incorporated into a nascent RNA strand, it can prevent RNA strand elongation and viral proliferation. Studies have also shown that the presence of purine analogs reduces favipiravir’s antiviral activity, showing competition between favipiravir-RTP and purine nucleosides for RdRp binding.
Although it was originally developed to treat influenza, the RdRp catalytic domain (favipiravir’s primary target), is expected to be similar for other RNA viruses. This conserved RdRp catalytic domain contributes to its broad-spectrum coverage.
Indication:
In 2014, it was approved in Japan to treat cases of influenza that were unresponsive to conventional treatment. Given its efficacy at targeting several strains of influenza, it has been investigated in other countries to treat novel viruses including Ebola and most recently, COVID-19.